Newsroom
New cells crucial for fingertip (digit tip) regeneration identified by Miller lab researchers
The ability to regrow a limb is a remarkable trait shared by few known species, and humans aren’t one of them. Researchers from the MSL’s Miller lab are striving to understand the science behind this amazing feat and find the key to regeneration which could hugely impact human health outcomes. Postdoctoral fellows Dr. Christine Eisner (she/her), Dr. Sruthi Purushothaman (she/her) and colleagues recently published an exciting paper in Cell Reports that sheds light on breakthrough discoveries in fingertip (digit tip) regeneration in mice.
“It’s long been known that the nail bed is essential for digit tip regeneration, but we haven’t understood why. We have answered this question in part with this paper. We’ve identified which cells within the nail mesenchyme, located beneath the nail and within the nail bed, contribute to regeneration of the digit tip,” Dr. Eisner explains.
Mesenchymal cells are found throughout most tissues and, depending on the environment, can acquire stem cell-like properties and contribute to the repair or scaring of various tissues upon injury. Through a combination of single cell transcriptomics and lineage tracing, the Miller lab has found a subset of mesenchymal cells called the “nail mesenchyme” that is labelled by the gene Lmx1b. Without the presence of these cells nothing regenerates and scar tissue forms.
“We knew there was something about the nail that was really important for regeneration. And now we’ve identified this population of cells which is essential for regrowth. Our discovery provides the basis for other research going forward to say ‘these are the cells that are important, why are they important?’,” says Dr. Purushothaman.
It was the salamander’s ability to regenerate lost limbs that first drew Dr. Purushothaman into this line of research. During her graduate studies Purushothaman worked on regeneration models with the axolotl, a species of salamander used in this line of research.
“To see an entire limb grow out is truly amazing. I wanted to better understand the underlying mechanisms that allowed these creatures to do this. Sadly, around the same time as I was doing this research, my mom went through a diabetic limb amputation. So, I started thinking of ways we could use the power of these cells to improve human health, ” Purushothaman says.
This deeply personal experience led Purushothaman to pursue further studies upon finishing her PhD, and she joined Dr. Miller’s lab in 2021. The idea of working on a mammalian model of regenerative research was very exciting. Dr. Eisner shares the same curiosity around tissue regeneration and enthusiasm for the promising findings of the Miller lab’s digit tip research model. She joined the lab in 2020 after completing a graduate program where she studied bone regeneration in mouse models at UBC’s School of Biomedical Engineering.
“I’ve always been interested to learn more about the human body and how we can grow and regenerate these different tissues. But sometimes we can’t. This research is helping us to understand how we can overcome these hurdles,” says Eisner.
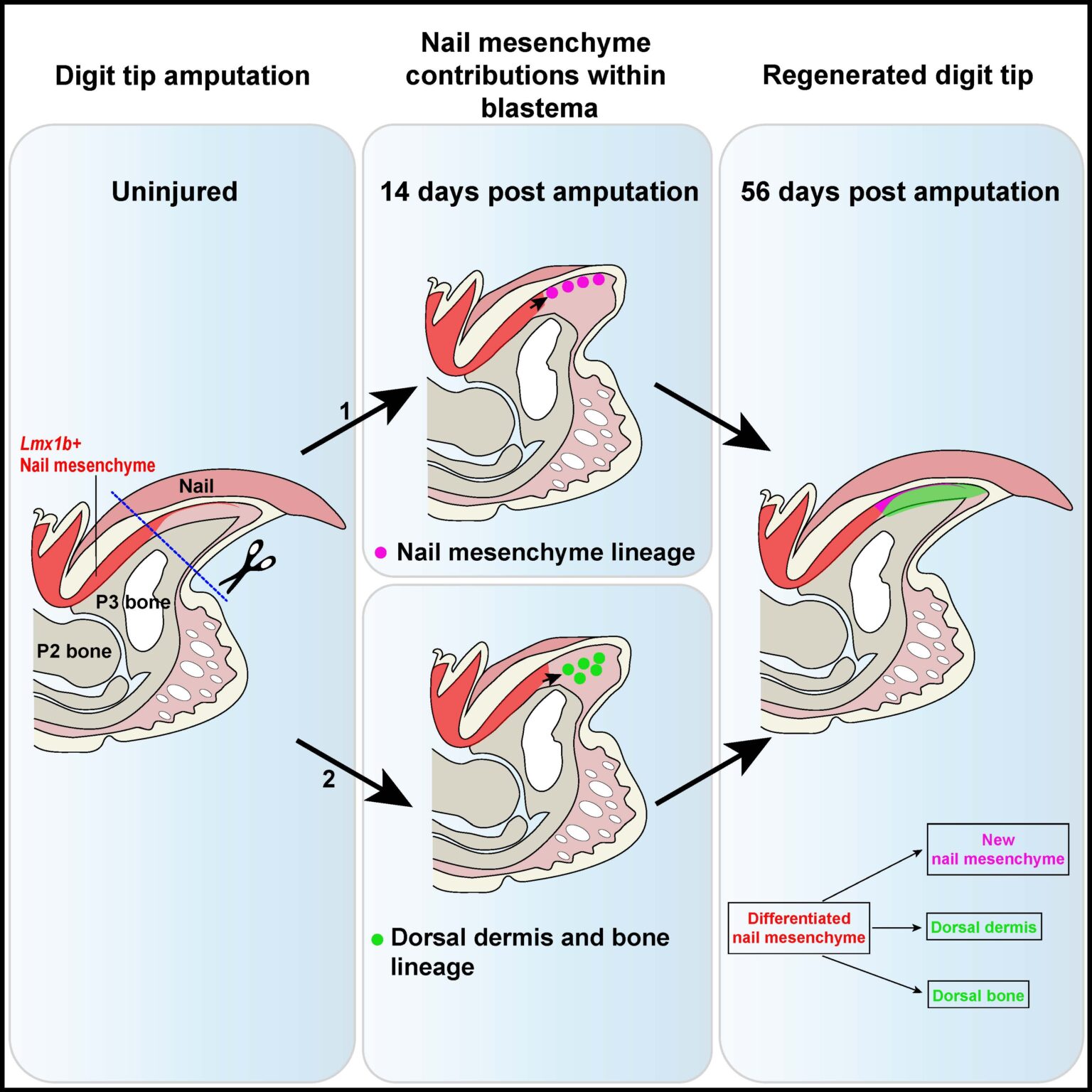
Lmx1b+ nail mesenchyme (labeled in red) contribute to and are indispensable for fingertip (digit tip) regeneration.
In vivo experiments with transgenic mice where nail mesenchyme cells have been destroyed, revealed that the amputations do did not regenerate, stressing the importance of these cells in successful regrowth. Eisner and Purushothaman will continue to explore different aspects of this research as they seek more answers about why these cells are needed for this process. One next step will be transplanting these cells to places where fibrosis would normally occur and seeing whether their presence changes the outcome. They are also examining the relationship between immune and mesenchymal cells, trying to uncover how they affect one another to create either a regenerative or non-regenerative environment.
“I think it’s really exciting that we have an answer to the question: What cells are important for regeneration? Now it’s a matter of defining why they are important and understanding how they function. If we could understand the molecular signals expressed by these cells and apply them to fibrosis situations to improve regenerative outcomes it would be a huge step forward in the field,” say Eisner.
Both researchers look forward to future applications of this knowledge. Leveraging the power of these regeneration cells could improve outcomes for burn victims, diabetic neuropathy sufferers and eventually patients facing amputations. The potentially positive impacts on human health are numerous and promising.
Quick links:
- Read the Cell Reports publication: Nail-associated mesenchymal cells contribute to and are essential for dorsal digit tip regeneration
- Read the Nature Research Highlight article on this research: Toes regrow with the help of these cells
- Learn more about the Miller Lab

